Half-Life Calculator
Calculate Radioactive Decay and Remaining Substance Mass
Please enter the required details and click Calculate.
Calculation Examples
📋Steps to Calculate
-
Enter the initial amount of the substance and its half-life value.
-
Enter the elapsed time, ensuring the time units match the half-life units (both in years, hours, or days).
-
Click "Calculate" to view the remaining amount, the fraction decayed, and the number of half-lives elapsed.
Mistakes to Avoid ⚠️
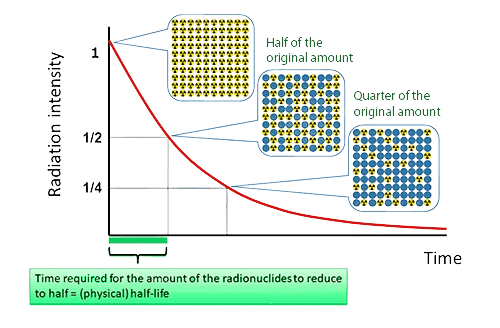
- Assuming a substance fully disappears after two or three half-lives. After two half-lives, 25% remains; after three, 12.5%. The quantity approaches zero asymptotically and never reaches it mathematically.
- Mixing time units between the half-life and elapsed time inputs, such as entering a half-life in hours but elapsed time in days, which overstates the number of half-lives by a factor of 24.
- Confusing half-life with mean lifetime (tau). Mean lifetime is the average time a single atom survives before decaying, equal to the half-life divided by the natural log of 2, approximately 1.4427 times the half-life.
Practical Applications📊
Calculate remaining radioactive activity in nuclear medicine isotopes such as Iodine-131 or Technetium-99m to determine safe handling windows and patient discharge timing.
Estimate residual drug concentration in the bloodstream for pharmacokinetics studies, therapeutic drug monitoring, and dosing interval planning.
Determine the age of organic materials in radiocarbon dating using Carbon-14, which has a half-life of approximately 5,730 years.