Pressure Converter Calculator
Convert between PSI, bar, kPa, MPa, atm, mmHg, and torr instantly. Uses NIST and BIPM-standard constants for automotive, industrial, and scientific pressure calculations.
Calculation Examples
📋Steps to Calculate
-
Enter the pressure value into the input field.
-
Select the source unit (e.g., bar, PSI, kPa, atm, mmHg).
-
Choose the target unit from the second dropdown.
-
Read the converted result instantly. For gauge pressure readings, add 1 atm to convert to absolute first.
Mistakes to Avoid ⚠️
- Confusing bar with millibar (mbar): 1 bar = 1,000 mbar. Entering a millibar reading into a bar field produces a result 1,000 times too large.
- Treating mmHg and torr as identical in high-precision work: they differ by about 0.000014% due to their different historical definitions. For clinical or metrological applications, use the correct unit explicitly.
- Ignoring gauge vs. absolute pressure: a tire inflated to 2.4 bar gauge is actually at 3.41 bar absolute at sea level. Using gauge values in absolute-pressure formulas (e.g., gas law calculations) gives incorrect results.
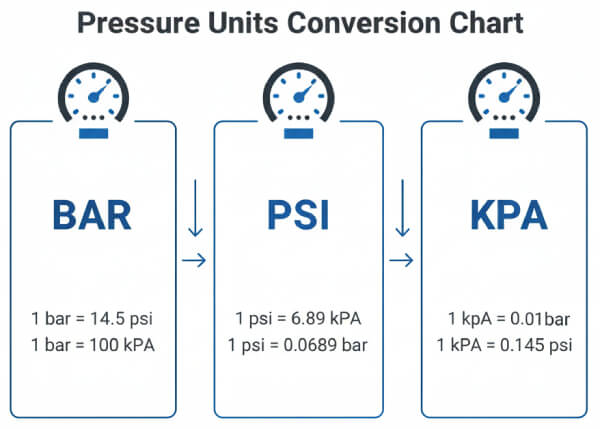
- Using the rounded bar-to-PSI factor of 14.5 in engineering calculations. The precise value is 14.50377. At pressures above 100 bar this rounding accumulates to several PSI of error.
Practical Applications📊
Automotive: Convert tire pressure between bar, PSI, and kPa. Most European vehicles specify pressure in bar; U.S. spec sheets use PSI. Correct conversion matters for fuel efficiency and tire longevity.
Industrial engineering: Translate hydraulic and pneumatic system specifications between MPa, PSI, and bar for equipment sourced from different countries.
Science and HVAC: Convert vacuum readings between mmHg, torr, and Pa for laboratory setups; convert refrigerant pressures between PSI and kPa for HVAC system diagnostics.
Meteorology: Convert weather station readings between hPa, mbar, atm, and inHg to normalize data across different instrument standards.